Pepose Vision is a patient-centered eye care practice with two locations in the St. Louis area, specializing in LASIK vision correction, cataract surgery, retina services, the Visian ICL, premium intraocular lens implants, (providing near, intermediate and distance vision) dry eye treatment and most any other vision care you may need. We offer the absolute latest technology, an advanced research center and a LASIK and cataract outpatient surgery center in our building. Most importantly, our team of experienced doctors and surgeons consistently achieve the very best possible outcomes for our patients.

We are proud to have been selected 25 years in a row as
“The doctors other doctors nationwide would choose for their own eye care and that of their families.”
We are able to help patients who are experiencing difficulty seeing at a distance with our signature, absolute latest technology LASIK Vision Correction, the Visian ICL Implantable Lens and contacts/glasses.
For patients that are experiencing trouble seeing up close, we have great solutions including the KAMRA Near Vision Inlay and of course, glasses and contacts.
We are experienced with the latest treatments, such as premium lens implants, that not only removes the cloudiness from cataracts but also improves vision, providing near, intermediate and distance vision.
We provide the absolute latest treatments for vision issues such as age-related macular degeneration and other retinal diseases, glaucoma, dry eyes, keratoconus, general eye care and much more.
LASIK is a popular surgery used to correct vision in people who are nearsighted, farsighted, orCloseup of Eye During LASIK Procedure have astigmatism. This non-invasive, bladeless surgery is quick and painless, and will improve your vision beyond your wildest dreams. You will enjoy great vision and no more hassles of glasses or contacts the same day as your surgery! Dr. Pepose participated in the initial FDA clinical trials to introduce LASIK, was the first to offer LASIK vision correction in the St. Louis region and continues to have the among the best track records for vision outcomes of any LASIK provider in the country. U.S. News & World Report has listed Dr. Pepose among the top 1% of LASIK surgeons nationwide. To learn more about LASIK and schedule your free consultation, please click below.

Blurry, cloudy vision from cataracts are a natural part of aging.Eye Doctors Performing Cataract Surgery in St Louis The good news is Pepose Vision can help. Our cataract surgery can not only help you see more clearly, but with our advanced premium lenses we can actually improve your vision. Often, you’ll be able to see better after your cataract removal at distances and close up and eliminate the need for glasses! We are a leader in the research of cataracts and in surgically implanting the latest, safest and most proven lens – right here in the convenience of the surgery center located in our building. Our surgeons are recognized annually as among the very best in the country. Click below to learn more about options to remove your cataracts and improve your vision, and to schedule an appointment.
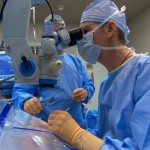
A healthy retina is necessary for good vision.Closeup of Eye Receiving Eye Drops During Retina Treatment Unfortunately, there are a number of retinal diseases that can affect vision, including age-related macular degeneration, diabetic retinopathy, flashes and floaters and more. The good news is Pepose Vision offers the latest surgical and medical treatment for retinal conditions. We are recognized as a leader in retinal care, and can provide you with the latest and most advanced methods of treatment in a customized plan to address these diseases and help improve your vision. Click below to learn more about our retinal services and how we can help, and to schedule your diagnosis.
Dr. Holekamp is the first in the bistate area to offer Susvimo, a novel drug delivery device that is surgically implanted and allows for continuous release of anti-VEGF, thereby reducing the frequency of intravitreal injections to only 2-3 times a year.
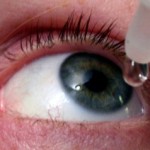
Dry eyes are very common, especially as the seasons change, but often not diagnosed nor treated.Woman Suffering from Allergies Dry eyes can not only effect your comfort and vision, but can become chronically worse. Pepose Vision specializes in the diagnosis and treatment of dry eyes. We offer the latest testing in house and continually run clinical trials in our research department to develop new and more effective treatments. We are experts in the LipiFlow® Thermal Pulsation System treatment. The good news is dry eyes can be treated. In fact, we often find patients suffer for years only to discover we can correct their issues quickly and easily. To learn more about dry eyes and how we can help, and to schedule an appointment, please click below.

Whatever your eye issue, odds are very good that we can diagnose and treat it. Pepose Vision takes pride in the knowledge, skills and experience of its doctors. Not only are we leaders in LASIK vision correction, cataract surgery, retinal services and dry eye treatment, we are also well-known as experts with glaucoma, keratoconus, corneal transplants, specialty contact lenses and pretty much any vision challenge you might face. We also offer thorough general eye care examinations to help diagnose potential issues early. To learn more about all the procedures we offer, please take a look at How May We Help You at the top of this page. To schedule an appointment for any vision need, please click below.
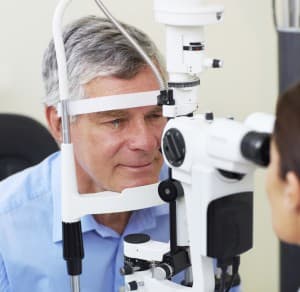


Dr. Pepose is included the Premier Surgeon 300, is a list of premium refractive cataract surgeons compiled by the editors and publisher of the influential Ocular Surgery News. He is identified as a leading innovator in the field of refractive cataract surgery and is one of only three surgeons selected in St. Louis.
The Achievement Award program recognizes individuals for their contributions to the Academy, its’ scientific and education programs, and to ophthalmology. The American Academy of Ophthalmology is the world’s largest association of eye physicians and surgeons with a global community of 32,000 medical doctors.

Millions suffer from Dry Eye, and yet the vast majority are not receiving the care they need. At our Accredited Dry Eye Center, you will be cared for by certified specialists in tear testing. We have the most advanced technology to diagnose and manage this irritating condition. Learn more about Dry Eye treatment.

The Tecnis Symfony® Intraocular Lenses provides a full range of continuous high-quality vision following cataract surgery, while also mitigating the effects of presbyopia by helping people focus on near objects. As a result, patients treated for cataracts not only have clearer vision, but improved vision at a range of distances, often reducing or eliminating the need for glasses.
Because so many patients are scheduling LASIK, we’ve added additional surgery slots and now have openings this month. Please visit our LASIK section to get more information and schedule your free, initial consultation where we’ll provide some key eye tests, explain the procedure in detail and answer your questions.
In addition to a wide range of advanced vision services, Pepose Vision is also a ophthalmological research center. We perform important clinical studies on new treatments for a variety of vision issues. Our studies offer our patients a chance to experience the latest breakthroughs in technology. And to play an important role in helping to develop future treatments. Please connect with Pepose Vision and see if there’s a clinical research study that could be right for you.
A new treatment has been approved by the FDA for treatment of diabetic macular edema (DME). ILUVIEN is an eye implant that automatically releases treatment for 36 months. Typically, patients being treated for DME require frequent, even monthly, injections into the eye. The real breakthrough with ILUVIEN is that a single injection lasts for 3 years. Please contact us to schedule an appointment to learn more.
We frequently offer free seminars where you can learn about the latest eye care technologies and opportunities.
These seminars include LASIK, KAMRA Inlay, Symfony and more. We will list current seminars here, and also send out a notification. If you aren’t on our email list, please scroll to the bottom of this page and sign up.
We accept most major insurance plans, plus medicare, and will help simplify your paperwork.
See what our patients have to say about their experience and results at Pepose Vision.
We’re also an advanced research center on the leading edge of the latest vision advances.
We offer flexible financing, including 0% plans, for procedures not covered by insurance.
Experienced Doctors
100%
customized treatment
100%
the latest technology
100%
best possible outcomes
100%
For credit-worthy applicants. 0% APR when paid in full in 60 months
-Patti









If you have any vision issues, your eyesight isn't what it used to be or you just haven't had an eye exam in a couple years, please come see us. Our experienced eye doctors and ophthalmologists will use advanced diagnostics to determine exactly how to help you see your very best, patiently answer your questions and carefully explain all your options.
call us